Soil carbon cycles and sequestration
Soil carbon storage is a vital ecosystem service, resulting from interactions of ecological processes. Human activities affecting these processes can lead to carbon loss or improved storage.
Posted on 14 Sep 2023

Fundamentals of Soil Organic Carbon
Soil organic matter is composed of soil microbes including bacteria and fungi, decaying material from once-living organisms such as plant and animal tissues, fecal material, and products formed from their decomposition. SOM is a heterogeneous mixture of materials that range in stage of decomposition from fresh plant residues to highly decomposed material known as humus. SOM is made of organic compounds that are highly enriched in carbon. Soil organic carbon (SOC) levels are directly related to the amount of organic matter contained in soil and SOC is often how organic matter is measured in soils.
SOC levels result from the interactions of several ecosystem processes, of which photosynthesis, respiration, and decomposition are key. Photosynthesis is the fixation of atmospheric CO2 into plant biomass. SOC input rates are primarily determined by the root biomass of a plant, but also include litter deposited from plant shoots. Soil C results both directly from growth and death of plant roots, as well as indirectly from the transfer of carbon-enriched compounds from roots to soil microbes. For example, many plants form symbiotic associations between their roots and specialized fungi in the soil known as mycorrhizae; the roots provide the fungi energy in the form of carbon while the fungi provide the plant with often-limiting nutrients such as phosphorus. Decomposition of biomass by soil microbes results in carbon loss as CO2 from the soil due to microbial respiration, while a small proportion of the original carbon is retained in the soil through the formation of humus, a product that often gives carbon-rich soils their characteristic dark color (Fig. 2). These various forms of SOC differ in their recalcitrance, or resistance to decomposition. Humus is highly recalcitrant, and this resistance to decomposition leads to a long residence time in soil. Plant debris is less recalcitrant, resulting in a much shorter residence time in soil. Other ecosystem processes that can lead to carbon loss include soil erosion and leaching of dissolved carbon into groundwater. When carbon inputs and outputs are in balance with one another, there is no net change in SOC levels. When carbon inputs from photosynthesis exceed C losses, SOC levels increase over time.

Photosynthesis, decomposition, and respiration rates are determined partly by climatic factors, most importantly soil temperature and moisture levels. For example, in the cold wet climates of the northern latitudes, rates of photosynthesis exceed decomposition resulting in high levels of SOC (Fig. 3). Arid regions have low levels of SOC mostly due to low primary production, while the tropics often have intermediate SOC levels due to high rates of both primary productivity and decomposition from warm temperatures and abundant rainfall. Temperate ecosystems can have high primary productivity during summer when temperature and moisture levels are highest, with cool temperatures during the rest of the year slowing decomposition rates such that organic matter slowly builds up over time (Fig. 4). While climatic conditions largely generate global patterns of soil carbon, other factors that vary on smaller spatial scales interact with climate to determine SOC levels. For example, soil texture — the relative proportions of sand, silt, and clay particles that make up a particular soil — or the mineralogy of those soil particles can have a significant impact on soil carbon stocks. Additionally, the processes of erosion and deposition act to redistribute soil carbon according to the topography of the landscape, with low-lying areas such as floodplains often having increased SOC relative to upslope positions.
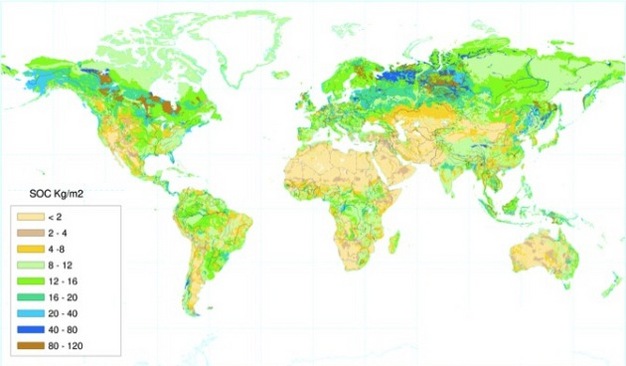

Soil Carbon and the Global Carbon Cycle
The amount of C in soil represents a substantial portion of the carbon found in terrestrial ecosystems of the planet. Total C in terrestrial ecosystems is approximately 3170 gigatons (GT; 1 GT = 1 petagram = 1 billion metric tons). Of this amount, nearly 80% (2500 GT) is found in soil (Lal 2008). Soil carbon can be either organic (1550 GT) or inorganic carbon (950 GT). The latter consists of elemental carbon and carbonate materials such as calcite, dolomite, and gypsum (Lal 2004). The amount of carbon found in living plants and animals is comparatively small relative to that found in soil (560 GT). The soil carbon pool is approximately 3.1 times larger than the atmospheric pool of 800 GT (Oelkers & Cole 2008). Only the ocean has a larger carbon pool, at about 38,400 GT of C, mostly in inorganic forms (Houghton 2007).
Published: 2021
Author(s): Todd A. Ontl & Lisa A. Schulte
